And you keep mischaracterizing what they said, even though it has been repeatedly pointed out and apparent to everyone.
And an op-ed piece centered on what was clearly a slip by Biden (in that it was not what he meant) is sorry evidence for your position.
BTW Operation Warp Speed was a complete failure. Where you living in this country at the end of last year?
Results 316 to 330 of 1196
Thread: All About Vaccines
-
05-17-2021, 10:07 AM #316
 There came a time when the Old Gods died! The Brave died with the Cunning! The Noble perished locked in battle with unleashed Evil! It was the last day for them! An ancient era was passing in fiery holocaust!
There came a time when the Old Gods died! The Brave died with the Cunning! The Noble perished locked in battle with unleashed Evil! It was the last day for them! An ancient era was passing in fiery holocaust!
-
05-17-2021, 10:14 AM #317

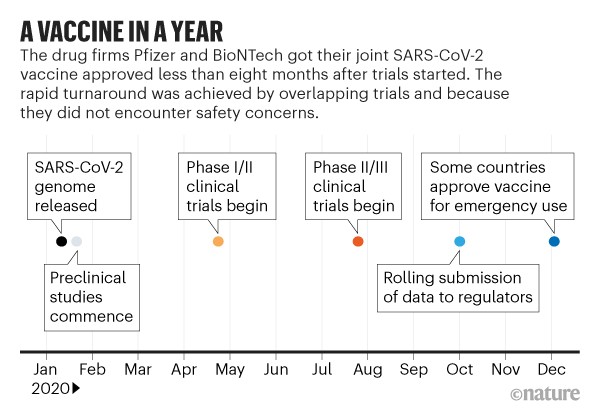
The most important of the vaccines so far seems to be BioNtech/Pfizer, which did not take Warp Speed money for its development, was finished first and was created by Muslim immigrant scientists in Germany.

They received the highest civil honor by the German government, the Bundesverdienstkreuz.
-
05-17-2021, 10:30 AM #318

The vaccine was developed by the end of Jan beginning of Feb 2020. Operation Warp Speed was not started until the end of March. It was purely a program to administer vaccines, not develop them. It was a failure.
U.S. vaccinations in 2020 fall far short of target of 20 million people
Only about 2.8 million Americans had received a COVID-19 vaccine going into the last day of December, putting the United States far short of the government’s target to vaccinate 20 million people this month.There came a time when the Old Gods died! The Brave died with the Cunning! The Noble perished locked in battle with unleashed Evil! It was the last day for them! An ancient era was passing in fiery holocaust!
-
05-17-2021, 12:22 PM #319Extraordinary Member

- Join Date
- Nov 2019
- Posts
- 6,134

This is one of the silliest statements and most completely wrong I've ever heard. I get you hate Trump, I'm not fond of the twit either, but his program to have vaccines ready before the end of the year worked, or you wouldn't be vaccinated, and like wouldn't for years, as that's what the skeptics said it would take. He also reserved 100s of millions of doses, spread across a number of candidates, guaranteeing the drug companies would make them money and give them incentive to work even if their effort didn't pan out. Biden did none of this. The fact is, you and I and every vaccinated person in this country is only so lucky because Trump tried something no one else would or did.
You DO read other country's media, right? The ones who are complaining that they didn't lock in those doses when we and Britain did. Most of the EU for example.
-
05-17-2021, 12:33 PM #320Extraordinary Member

- Join Date
- Nov 2019
- Posts
- 6,134

I don't know why you put up stuff that's SO easy to check when it is completely wrong.
Operation Warp Speed was NOT at all to do with getting shots into people's arms, (I don't know where you think those vaccines came from), it was about.....well, let's let Newsweek tell you:
https://www.newsweek.com/operation-w...pinion-1591018
And one final quote from them.Since distribution of the vaccines began on December 14, daily deaths and daily cases plummeted. By January 15, more than a million vaccine doses were being administered every day—even before President Biden took office. Over 150 million Americans have now received at least one dose of a vaccine. Thanks to OWS, the United States has administered the vaccine faster than any other nation in the world and has fully vaccinated more than twice as many people as the entire European Union combined.
I asked a high-level pharmaceutical executive directly involved in OWS if this program could have happened under a different administration. His answer was unequivocal: "No." The Trump administration was willing to take risks, create new precedents, cut Gordian knots and do whatever necessary to develop safe and effective vaccines in record time. Any other administration would likely have hidden behind bureaucratic delays, relied on doomsday predictions and generally moved at a much slower pace. In all likelihood, we would not have a vaccine today if not for President Trump's leadership in launching OWS.
BTW, it's easy to set a target below what your predecessor as President was already hitting. Biden, guy, stop being so transparent.
Another BTW, the first vaccine was approved for use on December 11th 2020, so if you wanted to try a shot without any testing, I suppose that's your choice, though I would urge against it. And of course you wouldn't have been allowed to get it.Last edited by achilles; 05-17-2021 at 12:52 PM.
-
05-17-2021, 01:09 PM #321Extraordinary Member

- Join Date
- Nov 2019
- Posts
- 6,134

Damn, ate most of my post. But, you are entirely wrong. Operation Warp Speed was to develop the vaccines and test them, It produced the first emergency use vaccine on December 11th 2020. Now if you wanted to test that vaccine in January, I wouldn't advise it, and you never would have been allowed. Plus, yes, Biden did set a goal of a million vaccines a day. Which was a pretty easy goal considering they were already doing over a million a day on inauguration day.
-
05-17-2021, 03:27 PM #322

Politely, no it wasn't. You actually posted an Op Ed from lying scumbag Tom Cotton and think it's true.
Operation Warp Speed was created at the end of March 2020. The Pfzer vaccine was developed in January 2020. Do the math.
The head of Operation Warp Speed admitted it failed.
https://www.statnews.com/2020/12/19/...ion-confusion/There came a time when the Old Gods died! The Brave died with the Cunning! The Noble perished locked in battle with unleashed Evil! It was the last day for them! An ancient era was passing in fiery holocaust!
-
05-17-2021, 04:53 PM #323Original join date: 11/23/2004
Eclectic Connoisseur of all things written, drawn, or imaginatively created.
-
05-17-2021, 05:04 PM #324

Operation Warp Speed: Accelerated COVID-19 Vaccine Development Status and Efforts to Address Manufacturing Challenges
Unveiling ‘Warp Speed,’ the White House’s America-first push for a coronavirus vaccine - May. 12, 2020Fast Facts
Operation Warp Speed was a federal effort that supported multiple COVID-19 vaccine candidates to speed up development.
We analyzed the program's vaccine candidates and found that their development followed traditional practices, with some adaptations. For example, some clinical trial phases overlapped with each other and with animal studies to accelerate development. Two vaccines were authorized for emergency use—our tracker
We also found that agencies are working to help mitigate vaccine manufacturing challenges. For example, agencies have taken steps to require contractors to prioritize vaccine production supplies.
Conventional wisdom is that a vaccine for COVID-19 is at least 1 year away, but the organizers of a U.S. government push called Operation Warp Speed have little use for conventional wisdom. The project, vaguely described to date but likely to be formally announced by the White House in the coming days, will pick a diverse set of vaccine candidates and pour essentially limitless resources into unprecedented comparative studies in animals, fast-tracked human trials, and manufacturing. Eschewing international cooperation—and any vaccine candidates from China—it hopes to have 300 million doses by January 2021 of a proven product, reserved for Americans.
Those and other details, spelled out for Science by a government official involved with Warp Speed, have unsettled some vaccine scientists and public health experts. They’re skeptical about the timeline and hope Warp Speed will complement, rather than compete with, ongoing COVID-19 vaccine efforts, including one announced last month by the National Institutes of Health (NIH). “Duplication only leads to infighting and slowing people down,” says Nicole Lurie, former U.S. assistant secretary for preparedness and response, who advises the Coalition for Epidemic Preparedness Innovations (CEPI), a nonprofit funding and helping coordinate COVID-19 vaccine efforts. “The U.S., and others around the world, should be engaged in this competition against the virus, not against one another.”New document reveals scope and structure of Operation Warp Speed and underscores vast military involvementWarp Speed, first revealed by Bloomberg News on 29 April, has so far only been outlined. President Donald Trump briefly discussed the initiative the next day, saying, “We’re going to fast track it like you’ve never seen before.” According to a CNN report on 1 May, which the source who spoke to Science confirmed, Warp Speed intends to deliver the first 100 million doses of a vaccine in November and another 200 million over the following 2 months.
An extraordinary 110 COVID-19 vaccines are in development, and eight candidates—four from Chinese companies—have entered small trials in people, according to an 11 May update from the World Health Organization (WHO). But there’s less than meets the eye in many of the efforts, says a vaccine veteran who asked not to be named. “Half of them are companies that have three guys, an administrative assistant, and a dog.”
The labyrinthine chart, dated July 30, shows that roughly 60 military officials — including at least four generals — are involved in the leadership of Operation Warp Speed, many of whom have never worked in health care or vaccine development. Just 29 of the roughly 90 leaders on the chart aren’t employed by the Department of Defense; most of them work for the Department of Health and Human Services and its subagencies.
Operation Warp Speed’s central goal is to develop, produce, and distribute 300 million doses of a coronavirus vaccine by January — and the military is intimately involved, according to Paul Mango, HHS’ deputy chief of staff for policy. It has already helped prop up more than two dozen vaccine manufacturing facilities — flying in equipment and raw materials from all over the world. It has also set up significant cybersecurity and physical security operations to ensure an eventual vaccine is guarded very closely from “state actors who don’t want us to be successful in this,” he said, adding that many of the Warp Speed discussions take place in protected rooms used to discuss classified information.One senior federal health official told STAT he was struck by the presence of soldiers in military uniforms walking around the health department’s headquarters in downtown Washington, and said that recently he has seen more than 100 officials in the corridors wearing “Desert Storm fatigues.”
“Military personnel won’t be familiar with the health resources available in a community,” said John Auerbach, CEO of Trust for America’s Health, a group closely aligned with public health departments. “They don’t know who the doctors are or where the community health centers are located or what resources they have. They don’t know where the pharmacies are. Public health people do know, that’s part of what they do.”Last edited by Tami; 05-17-2021 at 05:10 PM.
Original join date: 11/23/2004
Eclectic Connoisseur of all things written, drawn, or imaginatively created.
-
05-17-2021, 05:54 PM #325

One thing to remember about the Vaccines, this was an international endeavor. Other countries, like the UK, were pouring money into vaccine development as well. The companies that were making the vaccines didn't stand around waiting for a call from Washington D.C., they got on it right away knowing that there would be a lucrative international market for a successful COVID Vaccine.
Original join date: 11/23/2004
Eclectic Connoisseur of all things written, drawn, or imaginatively created.
-
05-17-2021, 06:48 PM #326BANNED

- Join Date
- Apr 2014
- Location
- New Hampshire
- Posts
- 4,641

This. Regardless of who was in office at the time some response would have been had to get vaccines developed and distributed in a timely manner. It's like giving Bush credit for bombing the Middle East after 9/11 (including the wrong country, because they had better military targets for CNN highlights and messed with his daddy). Do you believe Gore wouldn't have done the same? Just threw up his hands and said, "well we were attacked but whatcha gonna do?"
Trying to polish that turd's legacy by making him out to be a prescient and benevolent vaccine Santa after he did so much to undermine the response is laughable. Woodward quoted him early on as knowing how bad it was, but deliberately downplaying it. Then he decided to make masks a political issue rather than a necessary public health one. Then he pushed hydroxychloroquine as the focus of our efforts, despite experts telling him that was a mistake. Then he picked fights with experts like Fauci because the public rightly trusted and listened to them rather than Trump because this was serious and his word and expertise are dog s##t, and because they often had to put out fires when he'd have a press conference and say some wild bulls##t that had no bearing on reality and leave others who knew what they were talking about to come on and clean up his mess.
He sat back and let states take the initiative rather than having a Federally coordinated response because he didn't want the blame if things went wrong. His actions caused an untold number of deaths in this country. It may be tens of thousands, it may be hundreds of thousands, but make no mistake a lot of people are dead because he was either too incompetent or too afraid to do his job and that should never be forgotten. Otherwise terrible people are judged favorably by history when a moment comes and they rise to the occasion and do what is necessary. He wilted. Almost anyone else who was a serious contender for the Presidency wouldn't have let their ego get in the way of doing the job. The only silver lining, and again there are a lot of dead bodies so it's not saying much, is that he likely would have been a two-term President if he'd just handled the Covid response with basic competency.
-
05-17-2021, 09:19 PM #327Incredible Member

- Join Date
- Oct 2019
- Location
- New Jersey
- Posts
- 628

Hey , having read what you wrote about the former guy or other guy . You are absolutely right about it and Covid. There's only 2 good things to come out of all of this pandemic ****. One is that we have vaccines , especially the Pfizer which was invented in Germany and it's about 95% effective. The other is fortunately ( unfortunate for all the millions who died ) the former guy/other guy was not Elected or like you say, if he had handled the Covid response with basic competency he would have been a two-term President. I've been around for a long time and remember Watergate , but the former guy is the most corrupt , incompetent ever in my lifetime. Here's hoping that he does not run in 2024. Peace out !
-
05-17-2021, 09:22 PM #328Incredible Member

- Join Date
- Oct 2019
- Location
- New Jersey
- Posts
- 628

They deserve a Nobel and any other high honor that any country has !!!
-
05-18-2021, 12:11 AM #329

The sad thing about COVID is that is was allowed to become a political partisan issue when it should strictly be viewed as a health crisis.
I think the issue of lockdown made this inevitable.
(If I had a magic wand that would prevent any business from losing money or any person from being laid off from the pandemic, I can't help but think COVID deniers would be small in number)
The internet has also allowed fringe anti vaxxer conspiracy theories to become more accessible and creep into the mainstream. If your not trained in how to verify legitimate scholarly sources, it's easy to become victim to them.
Strange, when I was young anti vaxxer's were a far left eco thing, now it's a far right social Darwinist thing. In Canada, Alberta is the most right wing anti lockdown Province in the country, not surprisingly they are now leading North America in COVID cases.
-
05-18-2021, 01:10 AM #330
 Avatar: Here's to the late, great Steve Dillon. Best. Punisher. Artist. EVER!
Avatar: Here's to the late, great Steve Dillon. Best. Punisher. Artist. EVER!




 Reply With Quote
Reply With Quote